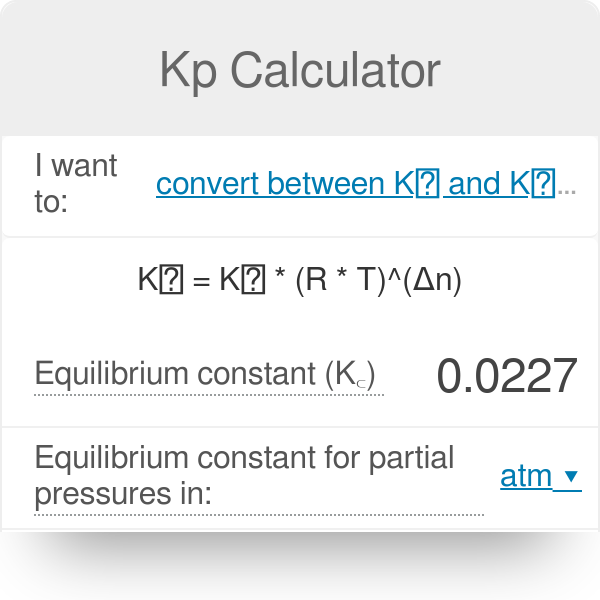
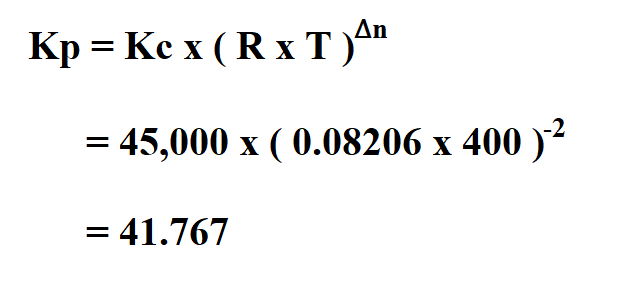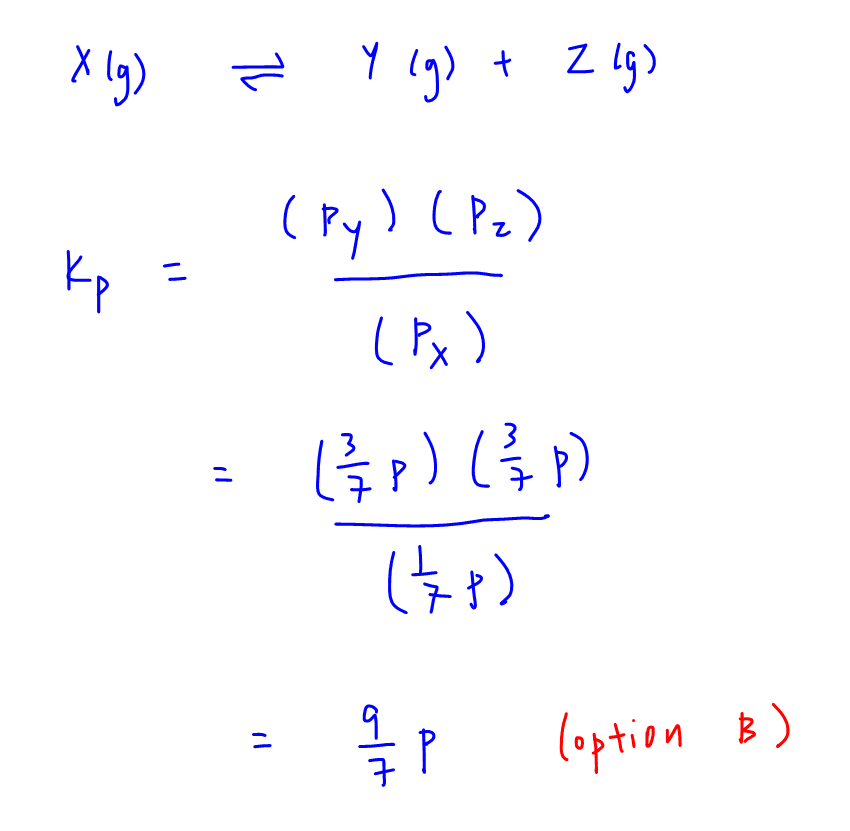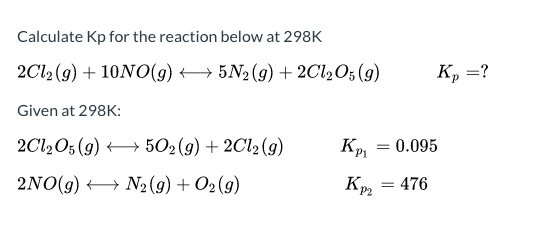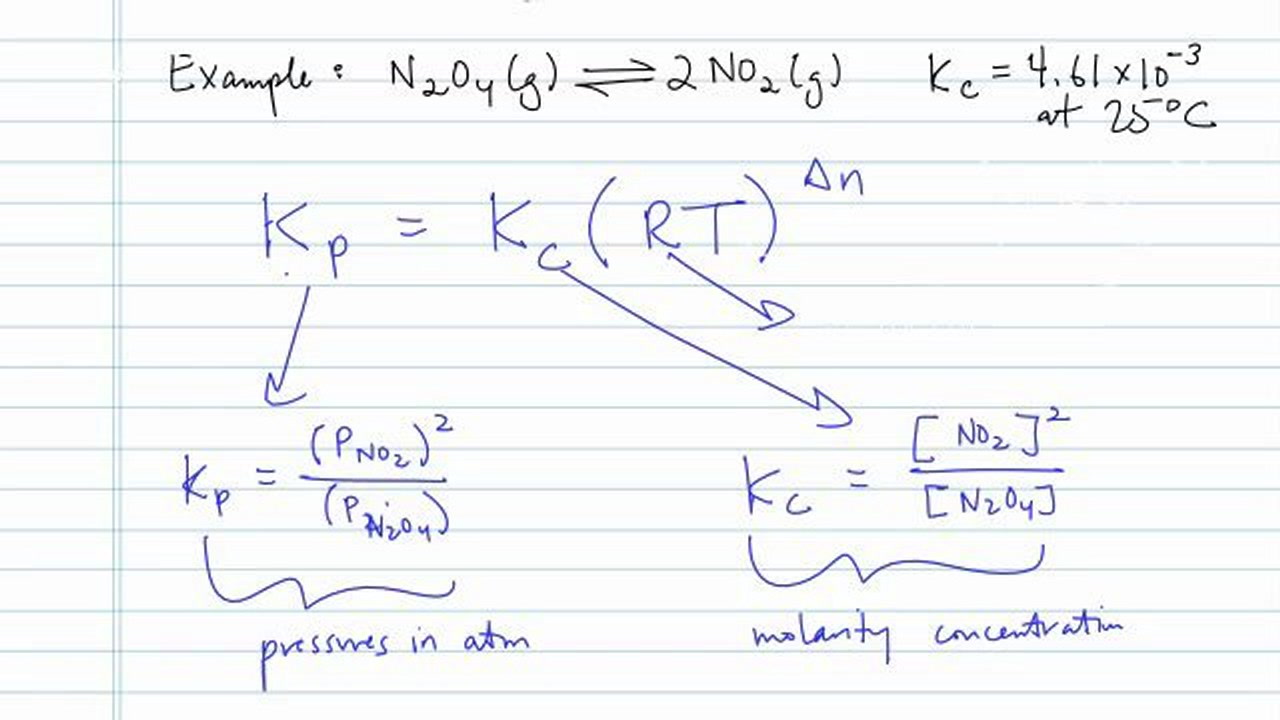Learn how the equilibrium constants Kc and Kp are related. | Chemistry lessons, Chemistry activities, Chemistry help

Learn how to calculate an equilibrium constant Kp. | Chemistry help, Chemistry education, Teaching chemistry

Learn how to calculate an equilibrium constant Kp. | Chemistry help, Chemistry education, Teaching chemistry
22. Calculate Kp for the equilibrium NH4HS (s) >NH3 (g)+H2S (g) if the total pressure inside the reaction vessel is 1.12 atm at 105^° C